Shop our range of products by category
ResMed CPAP machines and CPAP masks for sleep apnea treatmentFeatured Products
Popular Products
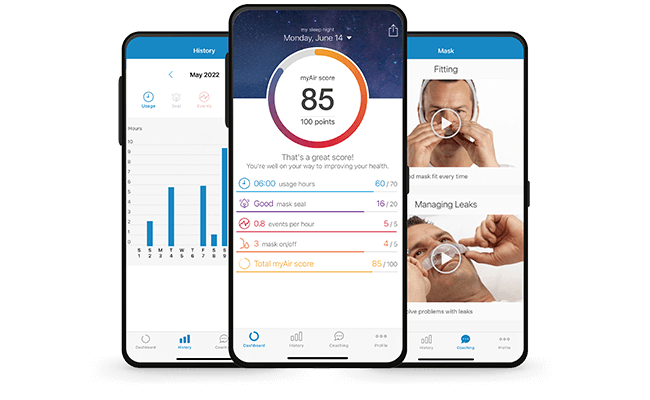
Track your PAP therapy with the myAir™ app
Wouldn’t it be motivating if you could see that you’re making progress on your PAP therapy treatment?
myAir can motivate you to stick with therapy by giving you insight into what’s going well and helping you to solve minor issues. Try the myAir app from ResMed, designed for Air10 PAP therapy devices.